In Principle:
Genetic Engineering involves the laboratory manipulation of DNA
What does a particular region of DNA do?
Isolation & manipulation of "gene of interest"
Genetic Engineering involves the laboratory manipulation of DNA
What does a particular region of DNA do?
Isolation & manipulation of "gene of interest"
in vivo cloning" [Nobel Prize 1980]
DNA sequencing [Nobel Prize 1980]
Polymerase Chain Reaction (PCR) [Nobel Prize 1993]
CRISPR-Cas9 [Nobel Prize 2020]
Bioinformatics
fresh viral, prokaryotic, or eukaryotic (plant & animal, etc.)
DNA separated from other molecules by selective binding
(CSHL animation of DNA extraction)
Ancient DNA (aDNA)
museum specimens: 10s ~ 100s years
fossils: 1,000s ~ 1,000,000s years
Ex.: insects in amber
Ex.: ancient humans: Neanderthals, Denisovans, & relatives
Forensic DNA
Forensics: data used as evidence
Ex.: blood / semen stains at crime scenes
Ex.: What species do these fillets come from?
Environmental DNA (eDNA)
Ex.: What taxa (prokaryotic phyla or eukaryotic species) present?
Marine, freshwater, terrestrial monitoring
In vivo Molecular Cloning of DNA
Type-II restriction endonucleases
cut DNA only at specific restriction (recognition) sites (list)
DNA palindrome -
"Able was I ere I saw Elba"
"Madam, I'm Adam"
"A man, a plan, a canal: Panama!"
"Straw? No! Too stupid a fad, I put soot on warts."
''Doc, note I dissent. A fast never prevents a fatness. I diet on cod."
"Saippuakivikauppias" - 15-letter Finnish word for a soap seller
restriction site reads the same in 5'

Overhanging TTAA-5' are "sticky ends"
Vector insertion
Vector - a means of moving DNA from one place to another
Plasmids - a circular, extrachromosomal DNA
"naked DNA", "bacterial virus"
pUC18 - an artificial plasmid with:
selectable markers that tell you when plasmid is present
antibiotic resistant (e.g., tetracycline or ampicillin)
lacZ gene produces beta-galactosidase,
metabolizes (unusual) Xgal sugar
lacZ gene includes polylinker
multiple, unique restriction sites
Recombinant DNA molecules formed when
linearization of vector DNA by digestion with endonuclease
ligation of "sticky ends" between source DNA & vector DNA
re-combines DNA from two different sources
In vivo cloning in E. coli
E. coli K12 strain
can't grow in presence of antibiotics (antibiotic-sensitive)
can't metabolize X-galactose (Xgal) sugars
Host transformation integrates plasmid DNA into bacterial chromosome
Bacterium acquires genes from plasmid, including "Gene of Interest"
Colony Selection Scheme: finding rare bacterium with recombinant DNA
Only E. coli cells with resistant plasmids grow on antibiotic medium;
only plasmids with functional lacZ gene can metabolize Xgal
lacZ(+)
lacZ functional
lacZ(-)
polylinker disrupts lacZ
Bulk bacterial culture of recombinant (white) colonies
Purify cloned plasmid DNA, cut cloned gene out with endonucleases:
Gene of Interest ready for Analysis
Polymerase
Chain Reaction
In vitro DNA "cloning": "DNA xeroxing" w/ four components & gadget
DNA template
anything with DNA in it
oligonucleotide primers ("oligos")
short (20 ~ 30 base) ssDNA complementary to gene of interest
some knowledge of gene is required :
"Universal primers" work across many species
Taq DNA polymerase
heat-stable enzyme from hot-spring bacteria (Thermus aquaticus)
functional at 70 ~ 80oC, withstands exposure to 95oC
dNTPs: four building-blocks for DNA
Thermal cycler: computer-controlled heating & cooling block
temperature change > 1oC / sec
PCR doubles gene copy number each cycle
denature / anneal / extend: 2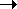 4
4 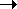 8
8 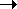 16
16 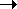 32
32  64 etc.:
64 etc.:
10 cycles = 210 = 103 copies, 20 cycles 106
copies, 30 cycles
106
copies, 30 cycles
 109 copies
109 copies
[ of PCR]
Thermal cycler automates process
replicates specific gene segment only
sufficient [DNA] for direct analysis
Variations: quantitative PCR (qPCR)
cf. qualitative (preparative) PCR produces DNA in bulk
quantitative (analytical) PCR rxns monitored in "real time"
Laboratory exercise in PCR primer design
In vitro DNA "cloning": "DNA xeroxing" w/ four components & gadget
DNA template
anything with DNA in it
oligonucleotide primers ("oligos")
short (20 ~ 30 base) ssDNA complementary to gene of interest
some knowledge of gene is required :
"Universal primers" work across many species
Taq DNA polymerase
heat-stable enzyme from hot-spring bacteria (Thermus aquaticus)
functional at 70 ~ 80oC, withstands exposure to 95oC
dNTPs: four building-blocks for DNA
Thermal cycler: computer-controlled heating & cooling block
temperature change > 1oC / sec
PCR doubles gene copy number each cycle
denature / anneal / extend: 2
10 cycles = 210 = 103 copies, 20 cycles
[ of PCR]
Thermal cycler automates process
replicates specific gene segment only
sufficient [DNA] for direct analysis
Variations: quantitative PCR (qPCR)
cf. qualitative (preparative) PCR produces DNA in bulk
quantitative (analytical) PCR rxns monitored in "real time"
Laboratory exercise in PCR primer design
Restriction mapping
Determining the order of & distances among restriction sites in DNA fragment:
provides an "outline" of the DNA sequence ["Mitochondrial Eve"]
(much) finer scale than three-point test cross chromosome map
Gel electrophoresis separates DNA fragments by molecular weight
( of agarose gel electrophoresis)
Fragment numbers & sizes compared in single & pairwise restriction digestions:
order & distances among sites determined as Restriction map
(CSHL animation)
Restriction maps of overlapping fragments assembled as contig map
Laboratory exercise in Restriction mapping
DNA sequencing
in vitro DNA replication copies one strand repeatedly
(cf PCR: both strands replicated)
Provides complete order of bases in a DNA fragment
DNA primer is complementary to 3' end of gene of interest
dideoxynucleotide terminators (ddNTPs)
stop strand growth during replication
four separate reactions terminate at ddA, ddC, ddG, or ddT
Sequencing gel shows series of partial DNA replications
Sequencing "ladder" autoradiogram read from bottom to top
( of dideoxy DNA sequencing)
Automated DNA sequencers uses laser fluorometry
ddNTPs attached to fluorescent dyes in single reaction
scanning laser activates & fluorometer "see" fluorescence colours (A C G T) [HOMEWORK]
computer "calls sequence" as chromatogram
( of automated DNA sequencing)
Next Generation sequencing uses massively-parallel, high-throughput methods
NextGen sequencers use capillary separation, other methods
Laboratory exercise with DNA Sequences
Southern Blot analysis
RFLP differences
Click here to go to the download site