Research Programs
Electrosynthesis at liquid|liquid interfaces
In this research program, the electrified liquid|liquid interface is exploited as a platform for the electrodless synthesis of nanocomposite thin-films.
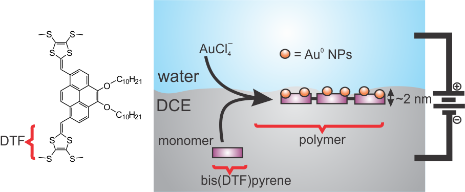
If the supporting electrolyte salts are sufficiently insoluble in either phase, than an electrode is immersed in either phase and the Galvani potential difference, localized across the water|oil or water|ionic liquid interface can be controlled externally and is polarized. The Galvani potential difference acts as the driving force for simple ion, facilitated ion, and electron transfer reactions.
References
[1] R. Moshrefi, K. Ryan, E.P. Connors, J.C. Walsh, E. Merschrod, G.J. Bodwell, T.J. Stockmann, Electrosynthesis of Au nanocluster embedded conductive polymer films at soft interfaces using dithiafulvenyl-functionalized pyrene, Nanoscale, 15 (2023) 5834-5842.
[2] R. Moshrefi, H. Przybyła, T.J. Stockmann, Simultaneous electro-generation/polymerization of Cu nanocluster embedded conductive poly(2,2′:5′,2′′-terthiophene) films at micro and macro liquid/liquid interfaces, Sci. Rep., 13 (2023) 1201.
[3] R. Moshrefi, T.J. Stockmann, Electrodeless Synthesis of Low Dispersity Au Nanoparticles and Nanoclusters at an Immiscible Micro Water/Ionic Liquid Interface, Nanomaterials, 12 (2022) 2748.
[4] R. Moshrefi, E.P. Connors, E. Merschrod, T.J. Stockmann, Simultaneous electropolymerization/Au nanoparticle generation at an electrified liquid/liquid micro-interface, Electrochim. Acta, 426 (2022) 140749.
[5] R. Moshrefi, A. Suryawanshi, T.J. Stockmann, Electrochemically controlled Au nanoparticle nucleation at a micro liquid/liquid interface using ferrocene as reducing agent, Electrochem. Commun., 122 (2021) 106894.
[6] M.D. Scanlon, E. Smirnov, T.J. Stockmann, P. Peljo, Gold Nanofilms at Liquid–Liquid Interfaces: An Emerging Platform for Redox Electrocatalysis, Nanoplasmonic Sensors, and Electrovariable Optics, Chem. Rev., 118 (2018) 3722-3751.
Single Entity Electrochemistry (SEE)
Single entity electrochemistry (SEE) is the described as monitoring or identifying single entities ‘one-at-a-time’ via an electroanalytical technique. ‘Entity’ in this case can be a single molecule, nanoparticle, protein, or even a living cell such as a bacteria. Indeed, the Coulter counter method developed in the 1950’s was one of the earliest electroanalytical methods. Most rely on chronoamperometry in which the potential is pulsed at a constant magnitude between two electrodes kept in separate chambers linked by a micro or nanopore while the ionic current is monitored over time, i.e., an i-t curve. In the case of the Coulter counter, bacteria transiting across a micro pore between two chambers disrupt the ionic current signal. In our work, stochastic impacts of metal and dielectric nanoparticles [1-3] at an ultramicroelectrode (UME) or nano-emulsions at a micro liquid/liquid interface [4] are also monitored in the i-t domain.
References
[1] N. Ahmadinasab, T.J. Stockmann, Single-Entity Electrochemical Detection of As-Prepared Metallic and Dielectric Nanoparticle Stochastic Impacts in a Phosphonium Ionic Liquid, ChemElectroChem, 9 (2022) e202200162.
[2] T.J. Stockmann, J.-F. Lemineur, H. Liu, C. Cometto, M. Robert, C. Combellas, F. Kanoufi, Single LiBH4 nanocrystal stochastic impacts at a micro water|ionic liquid interface, Electrochim. Acta, 299 (2019) 222-230.
[3] T.J. Stockmann, L. Angelé, V. Brasiliense, C. Combellas, F. Kanoufi, Platinum nanoparticle impacts at a liquid|liquid interface, Angew. Chem. Int. Ed., 56 (2017) 13493-13497.
[4] O.N. Egbe, B.H.P. Morrissey, N.E. Harvey, C. Schneider, L.S. Cahill, T. Jane Stockmann, Ionosome single entity electrochemical detection at a micro water/alkylphosphonium ionic liquid interface, J. Electroanal. Chem., 945 (2023) 117678.
Instrumentation
Heka PG-618-USB high-sensitivity potentiostat
