Stereoisometry
of
Amino
Acids

Stereoisometry
of
Amino
Acids
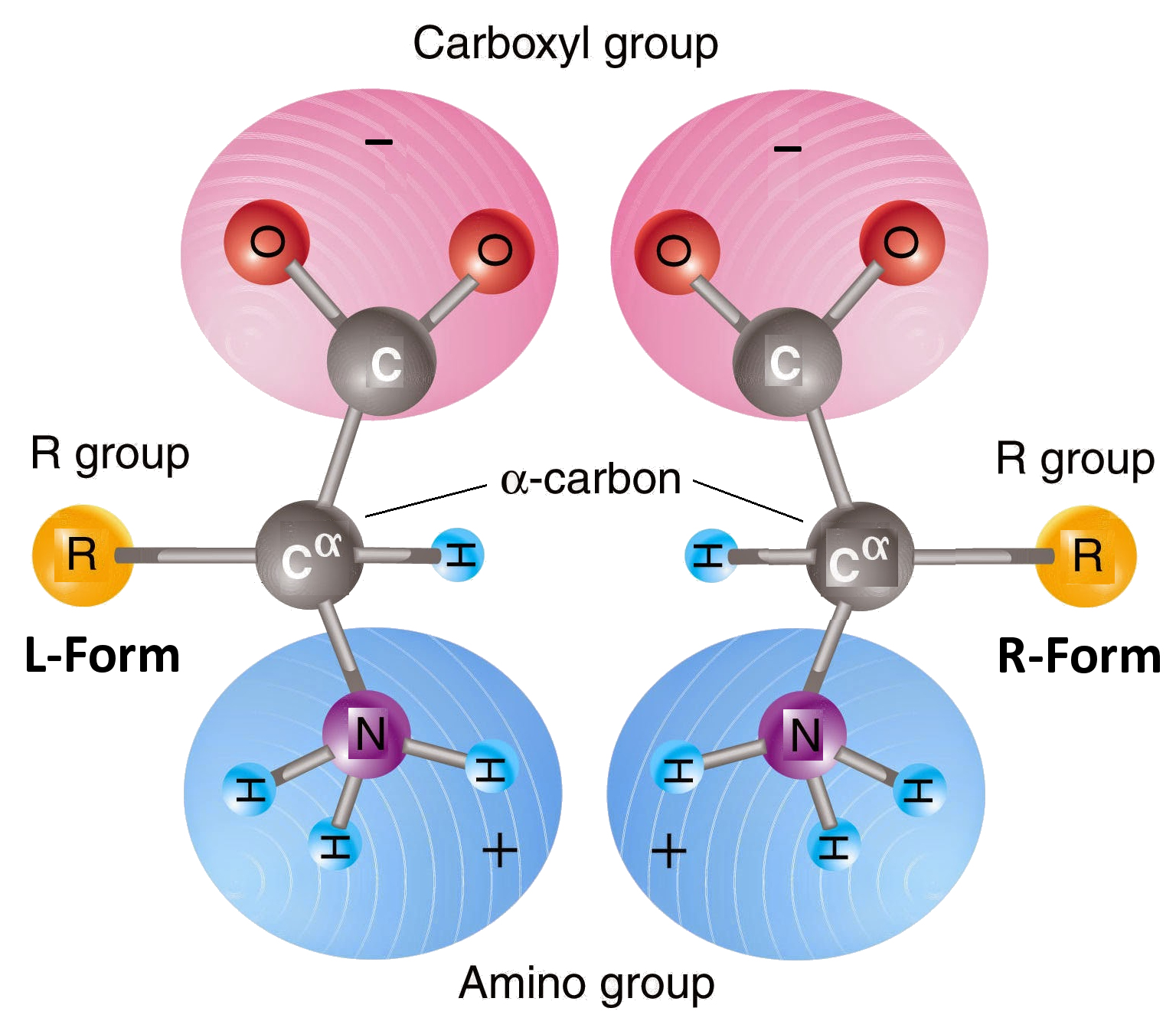
The four bonds of
the central (alpha) carbon
(C) of an amino acid are
directed towards the four corners of a tetrahedron. With respect to
the carboxyl (COOH) and amino (NH2) groups, there are two possible
arrangements of the H and Radical
group. These arrangement are mirror images of each other,
and are called stereoisomers
(enantiomers).
Stereoisomers are designated D (dextro-rotatory)
or L (levo-rotatory) according to the
direction in which the crystalline forms rotate polarized light,
to the right and left, respectively. All naturally-occurring
proteins comprise exclusively the L forms of amino acids.
If you
imagine holding the model at left with the COOH at the
bottom and the NH2 at the top, the R radical group is to the right,
in the D form. In the mirror image on the
right, the R group is on the left, in
the L form.
For an interesting science fiction treatments
of possible consequences of stereoisometry, see the 1969 film "Journey to
the Far Side of the Sun."